 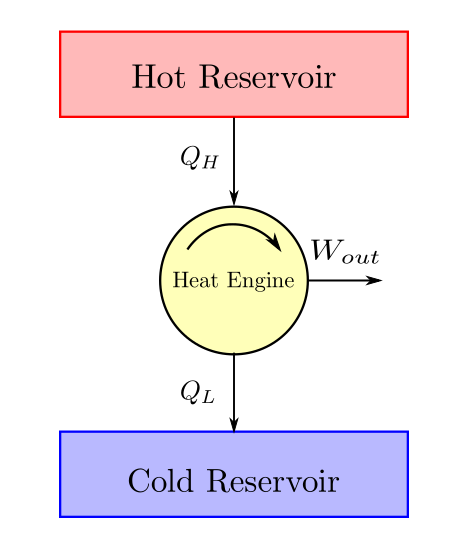
The reverse Carnot cycle is shown on a PV diagram in the following figure:Īs you can see in this figure, the cycle goes anticlockwise, because a heat pump requires work to operate (W<0). 4-1 Isothermal Compression: to close up the cycle, the ideal gas discharges an amount Q 1 of heat energy to the hot thermal reservoir.3-4 Adiabatic Compression: the ideal gas is then compressed adiabatically and its temperature rises back to the temperature of the hot thermal reservoir T 1.During this process, the gas absorbs a quantity Q 2 of heat from the cold thermal reservoir. 2-3 Isothermal Expansion: the ideal gas is allowed to expand at constant temperature in contact with the cold thermal reservoir at temperature T 2.1-2 Adiabatic Expansion: the ideal gas expands adiabatically, it cools down to the temperature T 2 of the cold thermal reservoir.The coefficient of performance (COP) of reversible or irreversible refrigerator or heat pump is given by. The reverse Carnot cycle consists of four reversible processes: A refrigerator or a heat pump that operates on the reversed Carnot cycle is called a Carnot refrigerator or a Carnot heat pump. As we will show below, it is the most efficient heat pump operating between two given temperatures.įirst, we will determine the coefficient of performance of the Carnot heat pump assuming that its working fluid is an ideal gas. A Carnot heat pump (or Carnot refrigerator) is a reverse Carnot heat engine, that absorbs heat from a cold thermal reservoir and transfers it to a warmer thermal reservoir. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
